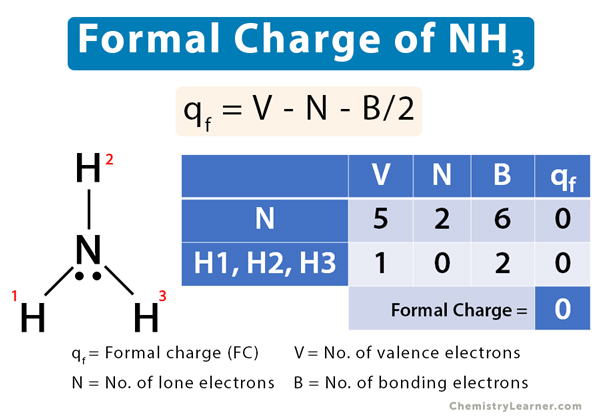
Step-by-step guide to calculating unionized (toxic) ammonia. UIA of... | Download Scientific Diagram

Calculate the equilibrium constant (Kc) for the formation of NH3 in the following reaction: N2 (g) + 3H2 (g) 2NH3 (g) At equilibrium, the concentration of NH3, H2 and N2 are 1.2 ×

Step-by-step guide to calculating unionized (toxic) ammonia. UIA of... | Download Scientific Diagram

stoichiometry - How to calculate the equilibrium constant for nitrogen, hydrogen, and ammonia? - Chemistry Stack Exchange
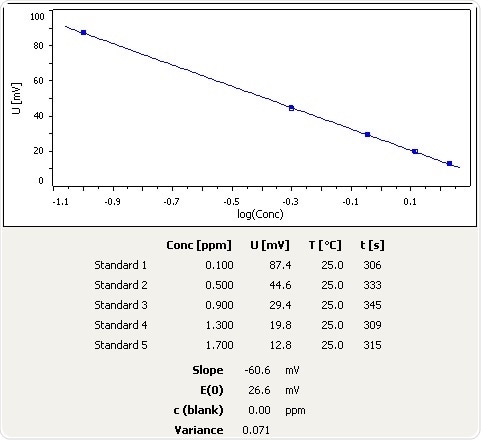
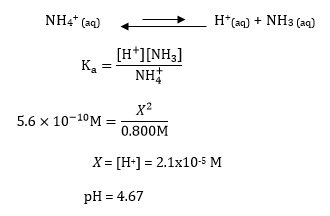
How to calculate your Un-ionized ammonia levels Information below collected from: Francis-Floyd, Ruth, Craig Watson, Denise Pett

Question Video: Calculating the Equilibrium Constant for Concentration Given the Initial Amount of Each Reactant | Nagwa

50 kg of nitrogen and 10 kg of hydrogen are mixed to produce ammonia. Calculate the ammonia formed and identify the limiting reagent in the production of ammonia in this situation.

SOLVED:The fertilizer, ammonium nitrate, is made by reacting ammonia with nitric acid. (Section 1.4) (a) Write a balanced equation, with state symbols, for the reaction of ammonia gas with nitric acid to
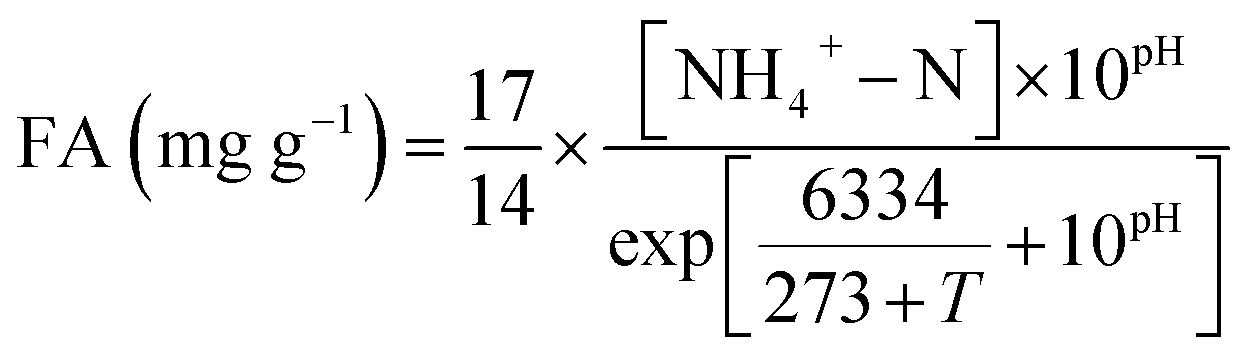
Characterizing the free ammonia exposure to the nutrients removal in activated sludge systems - RSC Advances (RSC Publishing) DOI:10.1039/C7RA10751J

Question Video: Calculating 𝐾_𝑝 at Equilibrium for a Mixture of Nitrogen, Hydrogen, and Ammonia | Nagwa

Ammonia Synthesis Production & Reaction | How Is Ammonia Made? - Video & Lesson Transcript | Study.com


![Ammonia [NH3] Molecular Weight Calculation - Laboratory Notes Ammonia [NH3] Molecular Weight Calculation - Laboratory Notes](https://www.laboratorynotes.com/wp-content/uploads/2023/03/ammonia-molecular-weight-calculation-300x150.jpg)